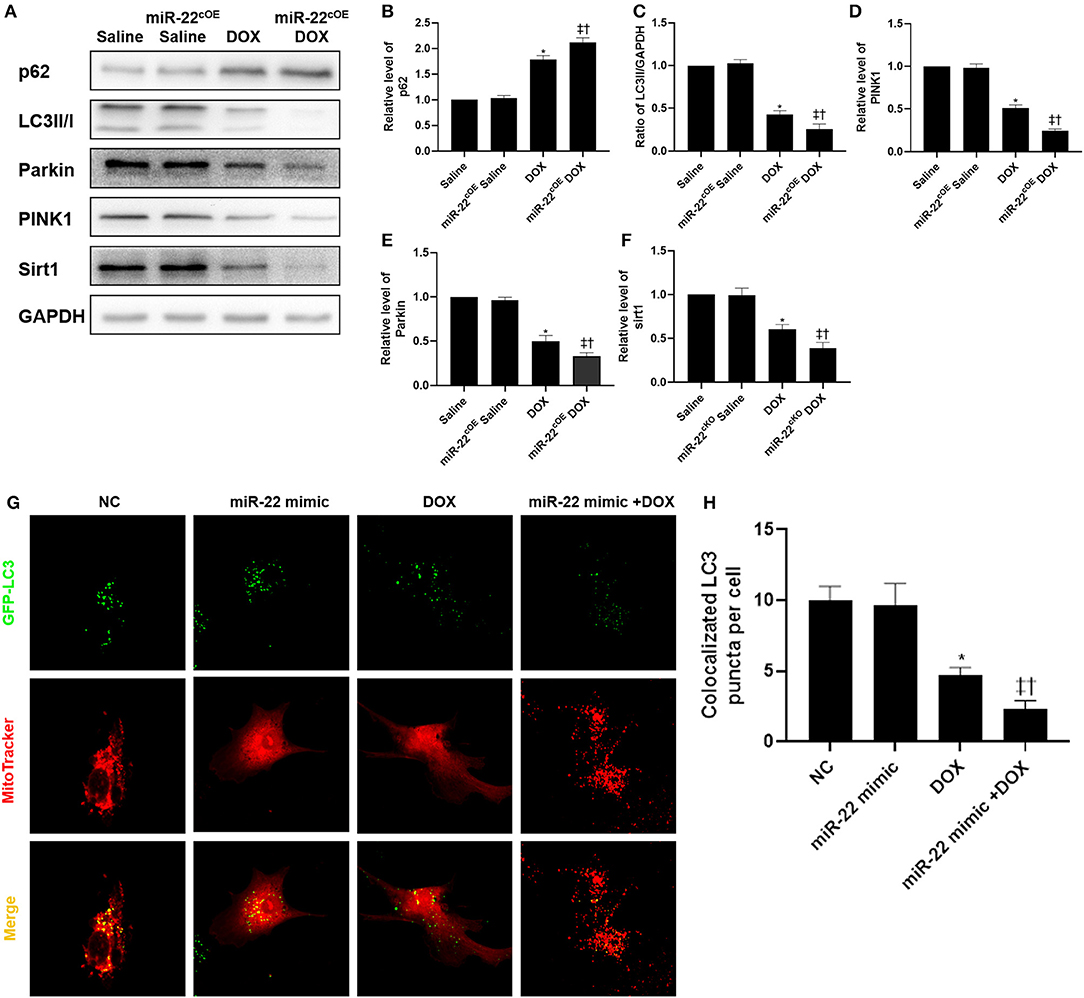
KLK11 has significant roles in physiological and pathological processes, including cancer biology. KLK11 is a member of the human kallikrein gene family. Accumulating evidence suggests that many kallikreins are implicated in carcinogenesis, and some have potential as novel markers for cancer and other diseases. Kallikreins (KLK) are a subgroup of serine proteases that have many physiological functions. Repression of mTOR with rapamycin can inhibit protein synthesis and repress cardiac hypertrophy induced by different pathological stress. Furthermore, we demonstrated that 5-hydroxytryptamine receptor 2A (HTR2A) fosters the development of cardiac hypertrophy by activating PI3K-PDK1-AKT-mTOR signaling. Besides, our current work demonstrated that Carboxypeptidase A4 promotes mTOR signaling during cardiomyocyte hypertrophy through activating PI3K-AKT signaling. For instance, AMP-activated protein kinase (AMPK) represses mTOR and protein synthesis via regulating the phosphorylation of the TSC1/TSC2 complex, the upstream inhibitor of mTOR. Within cardiomyocytes, mTOR can be regulated by various pathways. mTOR phosphorylated the ribosomal protein p70 S6K1 and 4EBP1 to facilitate de novo protein synthesis. In cardiomyocytes, protein synthesis is controlled by the mammalian target of rapamycin (mTOR) signaling pathway. Protein synthesis is one of the critical features of cardiomyocyte hypertrophy. However, the mechanisms underlying cardiac hypertrophy remain not fully understood. As thus, cardiac hypertrophy has been considered a promising target for the treatment of heart failure. These cardiomyocytes undergo hypertrophic growth to support the increase in demand in response to cardiac injury. Cardiomyocytes within the heart tissues are terminally differentiated cells unable to divide to support the cardiac injury. Cardiac hypertrophy is one of the primary mechanisms that participate in the development of cardiac dysfunction and heart failure. The morbidity and mortality of cardiac diseases and heart failure keep growing. Our findings demonstrated that KLK11 promoted cardiomyocyte hypertrophy by activating AKT-mTOR signaling to promote protein synthesis.ĭuring the last decades, cardiovascular diseases have become the leading cause of death worldwide. Besides, rapamycin treatment blocked the roles of KLK11 in the regulation of cardiomyocyte hypertrophy. Repression of mTOR with rapamycin blocked the effects of KLK11 on S6K1 and 4EBP1 as well as protein synthesis. Mechanism study demonstrated that KLK11 promoted the activation of AKT-mTOR signaling to promote S6K1 and 4EBP1 pathway and protein synthesis. We also observed that KLK11 promoted protein synthesis, the key feature of cardiomyocyte hypertrophy, by regulating the pivotal machines S6K1 and 4EBP1. Knockdown of KLK11 in mouse hearts inhibited TAC-induced decline in fraction shortening and ejection fraction, reduced the increase in heart weight, cardiomyocyte size, and expression of hypertrophic fetal genes. Besides, we knocked down KLK11 expression in mouse hearts with adeno-associated virus 9. Our in vitro experiments demonstrated that KLK11 overexpression promoted whereas KLK11 knockdown repressed cardiomyocytes hypertrophy induced by angiotensin II, as evidenced by cardiomyocyte size and the expression of hypertrophy-related fetal genes. We also induced cardiac hypertrophy in mice and observed the upregulation of KLK11 in hypertrophic hearts. The mRNA and protein levels of KLK11 were upregulated in human hypertrophic hearts. Gene expression was analyzed by quantitative real-time PCR. 
Protein synthesis was monitored by the incorporation of -leucine. The signaling pathway was analyzed by western blot. Cardiac function was analyzed by echocardiography. Mouse cardiac hypertrophy was induced by transverse aortic constriction (TAC), and cardiomyocyte hypertrophy was induced by angiotensin II. Human and mouse hypertrophic heart tissues were used to determine the expression of KLK11 with quantitative real-time PCR and western blot. Here we investigated the roles of Kallikrein 11 (KLK11) in cardiac hypertrophy. However, the underlying mechanisms of cardiac hypertrophy are not fully understood. Cardiovascular diseases have become the leading cause of death worldwide, and cardiac hypertrophy is the core mechanism underlying cardiac defect and heart failure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
